Reimagining catalyst layers: the case for ionomer-free iridium in PEMWE
Green hydrogen is set to play a pivotal role in the decarbonization of our energy systems. Proton exchange membrane water electrolysis (PEMWE) is among the most promising technologies for producing green hydrogen at scale. However, to meet the massive demand projected for the coming decades [1], PEM electrolyzer systems must be rapidly scaled to the gigawatt level, while simultaneously reducing costs and improving performance.
Currently, the catalyst layers in the systems’ electrodes are produced using ionomers. Ionomers are thought to enhance efficiency and stability in PEMWE by extending the catalytically active surface area and providing mechanical strength. However, there is a growing awareness that the ionomers’ presence may ultimately lower reaction rates and reduce electrolyzer lifetime when targeting lower catalyst loading. This article explores why adopting ionomer-free iridium catalyst layers is a key innovation for enabling the next generation of durable, high-performance electrolyzers that accelerate the transition to a net-zero energy system.
Iridium in PEM water electrolysis: a scarce but essential catalyst
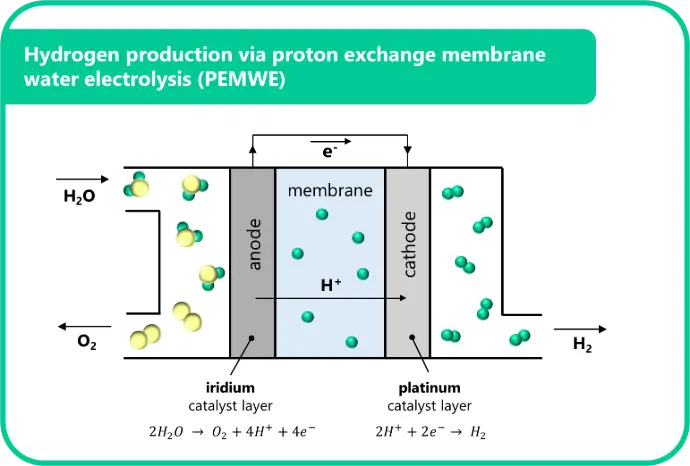
In a proton exchange membrane (PEM) electrolyzer, water is split into hydrogen and oxygen using electricity. The process is driven by electrochemical reactions at two electrodes (Figure 1) [2]. At the anode, water is oxidized to produce oxygen gas, protons, and electrons in the oxygen evolution reaction (OER). The protons migrate through the polymeric membrane to the cathode, where they combine with the electrons to form hydrogen gas via the hydrogen evolution reaction (HER).
The OER at the anode is kinetically sluggish and requires a highly active and stable catalyst, especially under the acidic, high-voltage conditions of the PEM cell. Iridium is currently the benchmark catalyst due to its exceptional stability and reasonable activity. However, iridium is one of the rarest and most expensive elements on Earth, making its efficient use a major focus in current PEM electrolyzer research [3].
The role of ionomers in conventional catalyst layers
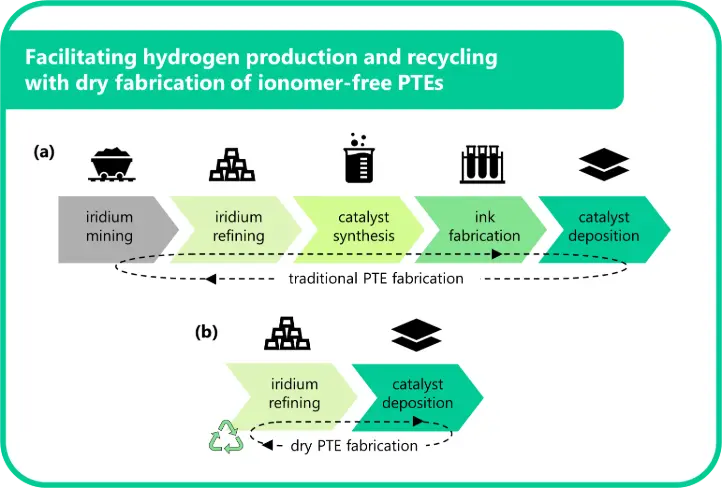
In PEM water electrolyzers, the catalyst is typically integrated into a membrane electrode assembly (MEA). In this configuration, the membrane is coated with the cathode and anode catalyst layers, and inserted between two porous transport layers (PTLs) (Figure 1). The anode catalyst layer – the focus of this article – is commonly prepared by first formulating an ink consisting of iridium particles, ionomer, and solvent. The ink is then uniformly applied to the membrane using wet deposition techniques such as spray coating or slot-die coating [4]. Once the solvent evaporates, a cohesive layer of iridium and ionomer remains, forming the active surface where the OER occurs. An alternative electrolyzer fabrication approach involves coating the catalyst layers directly onto the PTLs instead of the membrane, resulting in what is known as porous transport electrodes (PTEs).
Traditionally, the inclusion of ionomer in the catalyst layer has been considered crucial because the ionomer’s negative charges provide ionic conductivity, ensuring that protons generated at the anode can efficiently migrate toward the electrolyzer membrane. Additionally, the ionomer helps bind the catalyst particles to each other and to the support structure, contributing to the mechanical integrity of the assembly. However, it is postulated that the use of ionomers also suffers from a critical challenge: they are known to provide a physical barrier to oxygen diffusion during PEMWE – especially at high currents, where oxygen bubble formation is extremely intense [5]. Therefore, to maximize hydrogen production efficiency, the catalyst loading and ionomer content need to be carefully balanced.
Four key benefits of ionomer-free iridium layers
What if ionomers, once considered essential to PEM electrolyzer performance, aren’t as critical as we thought? This idea is gaining traction in the scientific community, with more and more studies exploring ionomer-free catalyst layers and challenging long-held assumptions in PEM electrolyzer design. As the field races to meet ambitious long-term electrolyzer capacity targets, ionomer-free PTEs are emerging as not just a viable alternative to their ionomer-containing counterparts, but potentially even a superior option. Below, we outline four key advantages that make ionomer-free iridium catalyst layers especially compelling.
1) Enabling efficient iridium recycling
To meet capacity growth goals, the limited global supply of iridium must be used more efficiently in electrolyzer stacks. While improved catalyst designs help maximize iridium utilization, recycling from end-of-life electrolyzer components is crucial to avoid future supply bottlenecks. Studies suggest that with proper recycling systems in place, iridium scarcity may no longer constrain PEMWE scale-up [6]. However, iridium recovery from PTEs remains underdeveloped and needs more attention to secure long-term material availability.
The presence of ionomers in the catalyst layers forms a major obstacle in recycling iridium from conventional electrodes. The most widely used ionomer, Nafion, is a fluorinated polymer that belongs to the class of PFAS (per- and polyfluoroalkyl substances), known for their chemical stability and environmental persistence. Nafion not only complicates the selective recovery of iridium but can also produce extremely hazardous gases when processed, especially during high-temperature or oxidative recycling steps [7]. In contrast, ionomer-free PTEs offer a cleaner and more accessible platform for recycling, paving the way for safer and more efficient recycling solutions.
2) Simplifying production by eliminating ink-based fabrication steps
Ionomer-free iridium catalyst layers are produced using dry deposition techniques that greatly simplify the PTE fabrication process. Conventional methods rely on the wet deposition of catalyst inks, which require formulation optimization steps and introduce challenges such as solvent flammability, sensitivity to fabrication parameters, and variability in quality [8]. Dry deposition of iridium catalyst layers on PTLs eliminates the need for inks, removing solvent handling and drying steps from the fabrication process.
3) Boosting electrolyzer efficiency
Ionomer-free iridium catalyst layers have demonstrated improved performance compared to conventional ionomer-containing electrodes, particularly when used at industrially relevant high current densities. For example, Lee et al. reported that ionomer-free PTEs outperformed traditional catalyst-coated membranes at current densities above 2 A·cm², with continued advantage up to 10 A·cm² under 3 V operation [7]. Similarly, Wang et al. demonstrated nearly 90 mV lower cell voltage at 6 A·cm² compared to a commercial baseline, despite using no ionomer in either electrode [9].
The performance improvements can be attributed to the elimination of the ionomer phase. The ionomer phase is not electrically conductive and can impede electron mobility within the catalyst layer. Moreover, the presence of ionomer provides a physical barrier to oxygen diffusion, leading to the formation of oxygen bubbles that further hinder mass transport during PEMWE. Finally, the ionomers or their degradation products can interfere with the catalyst’s active sites, reducing its ability to facilitate
OER. It is likely that a combination of these factors leads to inefficient catalyst utilization and ultimately limits the electrolyzer performance.
4) Enhancing durability for prolonged electrode lifetimes
Ionomer-free iridium catalyst layers have also shown remarkable improvements in durability compared to traditional ionomer-containing electrodes. Lee et al. compared the durability of ionomer-containing and ionomer-free iridium layers in an accelerated stress test (AST) [7]. For the ionomer-free layers, the researchers measured a degradation rate of just 0.58 mV per 1000 cycles at a current density of 4 A·cm²: nearly ten times lower than the rate reported for its ionomer-containing counterpart operated at 2 A·cm². The same study also demonstrated successful long-term operation of the ionomer-free electrodes, maintaining stable performance for over 400 hours with degradation rates comparable to the best values reported in literature.
This enhanced durability is likely due to the fact that the ionomer can degrade under high voltage and oxygen-rich conditions, leading to structural instability and failure. By eliminating the ionomer, ionomer-free iridium layers reduce the need for frequent component replacements, ultimately lowering system costs and minimizing iridium consumption over time.
Nanoprinting ionomer-free nanoporous iridium layers
VSParticle’s nanoprinting technology enables the fabrication of high-quality, ionomer-free iridium layers with a nanoporous structure ideally suited for PEM water electrolysis. Using a dry method based on spark ablation, the VSP-P1 Nanoprinter directly deposits iridium nanoparticles onto PTLs without the need for solvents, binders, or ionomers. This results in ultra-pure, structurally uniform catalyst layers with a high electrochemically active surface area. These layers have the potential to serve as next-generation anodes for PEM electrolyzers: by delivering high performance, enhanced durability, and facilitating iridium recycling, they support scalable and sustainable hydrogen production at lower cost.
-----------------
References
[1] International Energy Agency (IEA). “Global Hydrogen Review 2024” (2024). https://www.iea.org/reports/global-hydrogen-review-2024
[2] K. Zhang, X. Liang, L. Wang, K. Sun, Y. Wang, Z. Xie, Q. Wu, X. Bai, M.S. Hamdy, H. Chen, and X. Zou. “Status and perspectives of key materials for PEM electrolyzer.” Nano Research Energy, 1(3) (2022):, e9120032. doi.org/10.26599/NRE.2022.9120032
[3] C. Minke, M. Suermann, B. Bensmann, and R. Hanke-Rauschenbach. "Is iridium demand a potential bottleneck in the realization of large-scale PEM water electrolysis?" International Journal of Hydrogen Energy 46(46) (2021): 23581-23590. doi.org/10.1016/j.ijhydene.2021.04.174
[4] J. Park, Z. Kang, G. Bender, M. Ulsh, and S.A. Mauger. “Roll-to-roll production of catalyst coated membranes for low-temperature electrolyzers”. Journal of Power Sources, 479 (2020): p.228819. doi.org/10.1149/1945-7111/abc746
[5] M. Chen, Y. Ren, C. Lee, and J.K. Lee. “Ionomer-free electrodes in PEM water electrolyzers: a critical review”. (2025) Journal of Physics: Energy. doi.org/10.1088/2515-7655/adf6e3
[6] Johnson Matthey. “Two key focus areas will ensure iridium availability does not stall electrolyser growth” (2022). https://matthey.com/documents/161599/0/JM+Iridium+White+Paper.pdf/db45aebe-8182-2fc1-548a-a3072a4e6423?t=1669022842596
[7] J.K. Lee, G. Anderson, A.W. Tricker, F. Babbe, A. Madan, D.A. Cullen, D.A., Arregui-Mena, J.D., Danilovic, N., Mukundan, R., Weber, A.Z. and Peng, X., 2023. Ionomer-free and recyclable porous-transport electrode for high-performing proton-exchange-membrane water electrolysis. Nature Communications, 14(1), p.4592. doi.org/10.1038/s41467-023-40375-x
[8] H. Liu, L. Ney, N. Zamel, and X. Li. “Effect of catalyst ink and formation process on the multiscale structure of catalyst layers in PEM fuel cells”. Applied Sciences, 12(8) (2022): p.3776. doi.org/10.3390/app12083776
[9] W. Wang, L. Ding, J. Li, Z. Xie, M.M. Mench, and F.Y. Zhang. “An ink-free integrated dual electrode assembly for economical proton electrolyzer membrane water electrolysis at ultrahigh current densities”. Chemical Engineering Journal, 494 (2024): p. 153015. doi.org/10.1016/j.cej.2024.153015